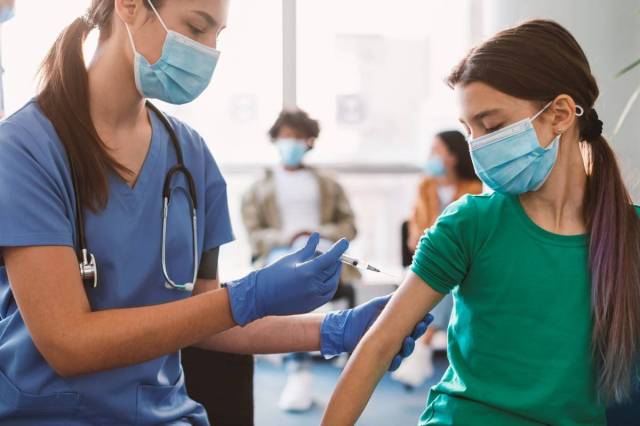
As Omicron continues to make a comeback, there is welcome news on the horizon. The Food and Drug Administration has just authorized booster shots for the Pfizer vaccine for kids ages five to 11.
Pfizer has been hard at work, requesting approval for its two-shot dose for the under-five set back in February. Now, the children who received their set of shots when it became available in November can top off their waning immunity with the booster. While the FDA has already authorized the new shot, the CDC is still planning to meet with a panel of outside experts on Thursday before officially recommending it, per The New York Times.
“The FDA has determined that the known and potential benefits of a single booster dose of the Pfizer-BioNTech COVID-19 Vaccine for children 5 through 11 years of age at least five months after completing a primary series outweigh its known and potential risks and that a booster dose can help provide continued protection against COVID-19 in this and older age groups,” says Peter Marks, M.D., Ph.D., director of the FDA’s Center for Biologics Evaluation and Research.
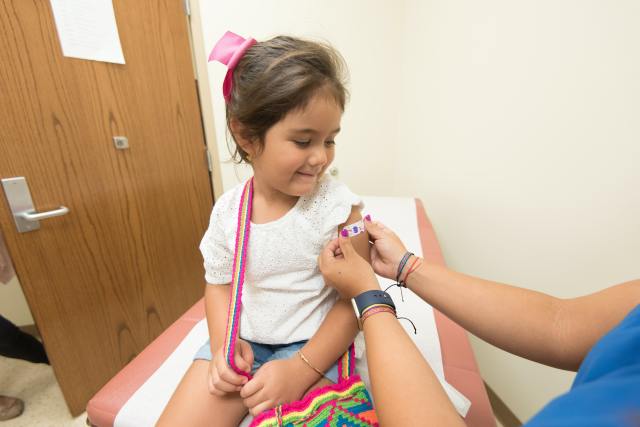
Kids will be eligible to receive the booster at least five months after their second dose. With the vaccine coming out in November 2021, those who got it immediately are already due for the booster; unfortunately, that number is not as high as it should be.
While more than eight million children have completed the two-shot vaccine, that’s far less than the 28 million that make up the eligible age group. In total, it means just 29% have received two doses and 6% just one dose. With a low turnout for the initial vaccine, booster acceptance may be an issue, as clearly parents are wary in the first place.
The booster shot is the same strength as the initial two doses, but there is no official news on when parents can sign their children up to receive it just yet. Make sure to check with your pediatrician before making your appointment.
RELATED STORIES
Pfizer Asks FDA to Approve 2-Dose Vaccine for Kids Under 5
Moderna Asks FDA for Covid-19 Vaccine Authorization for Kids 5 & Under
Expert Advice about COVID-19 Vaccines and Safety for Kids